Medwatch Form 3500A - Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected.
Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be.
An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day:
FDA MedWatch 3500A (August 2024 Version) Generation Mapping Veeva
Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may.
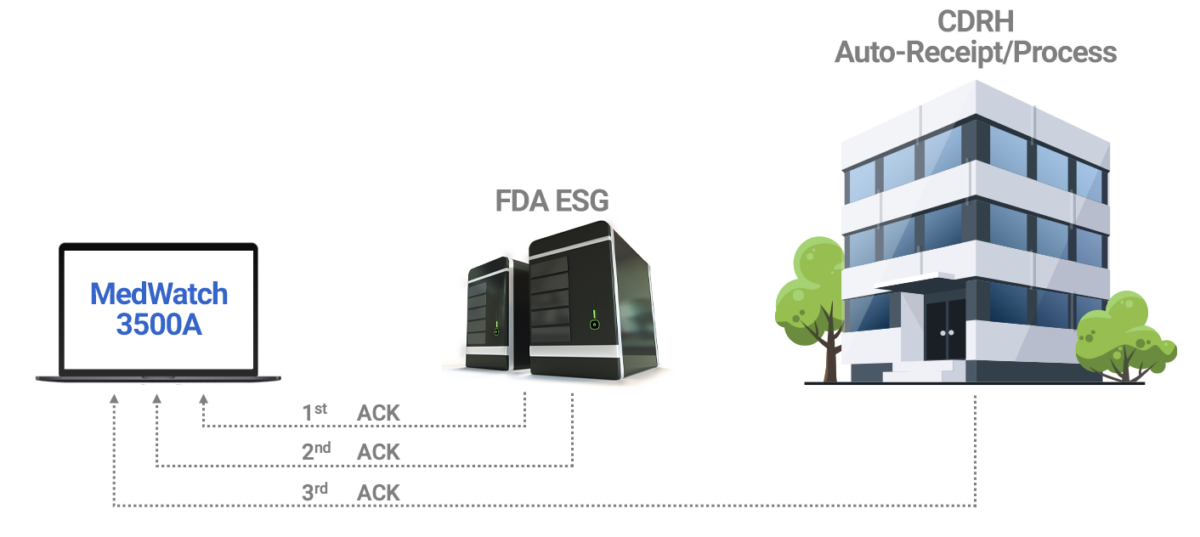
eMDR Solution Automates Submissions for MedWatch 3500A AssurX
An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional).
FDA MedWatch 3500A (August 2024 Version) Generation Mapping Veeva
Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be..
PPT Adverse Event Reporting at FDA PowerPoint Presentation, free
Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: An important fda program called medwatch allows health care professionals and consumers to report serious problems that.
SOLUTION Fda Form 3 Karen Studypool
Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or.
PPT Clinical Analysis of Adverse Drug Reactions PowerPoint
Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: An important fda program called medwatch allows health care professionals and consumers to report serious problems that.
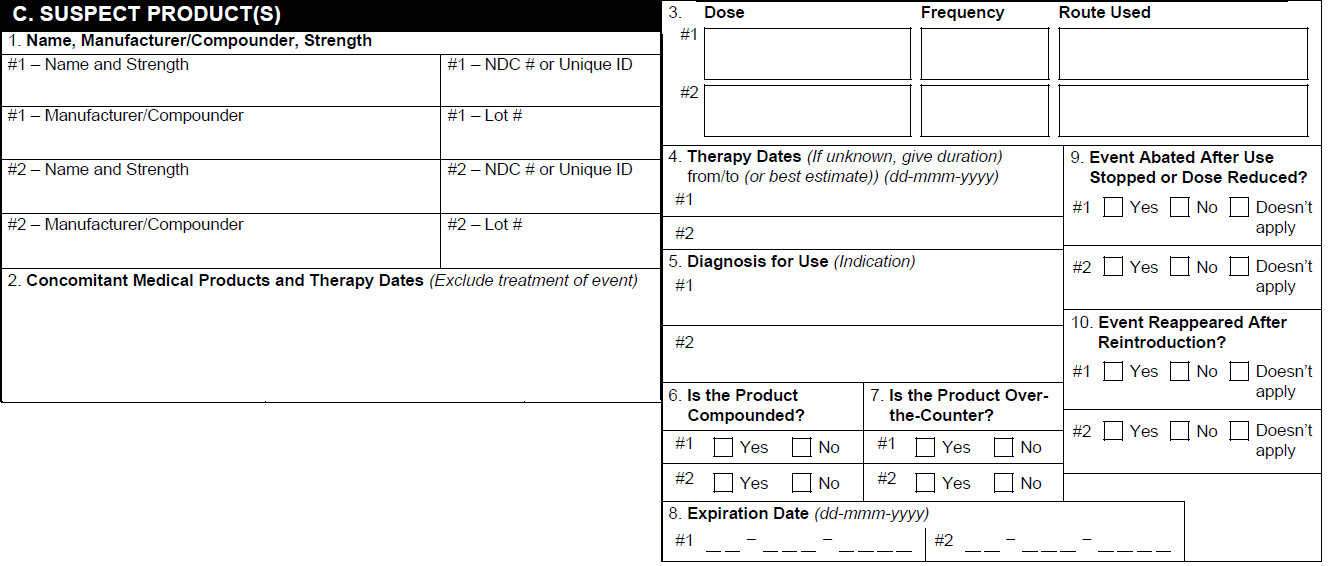
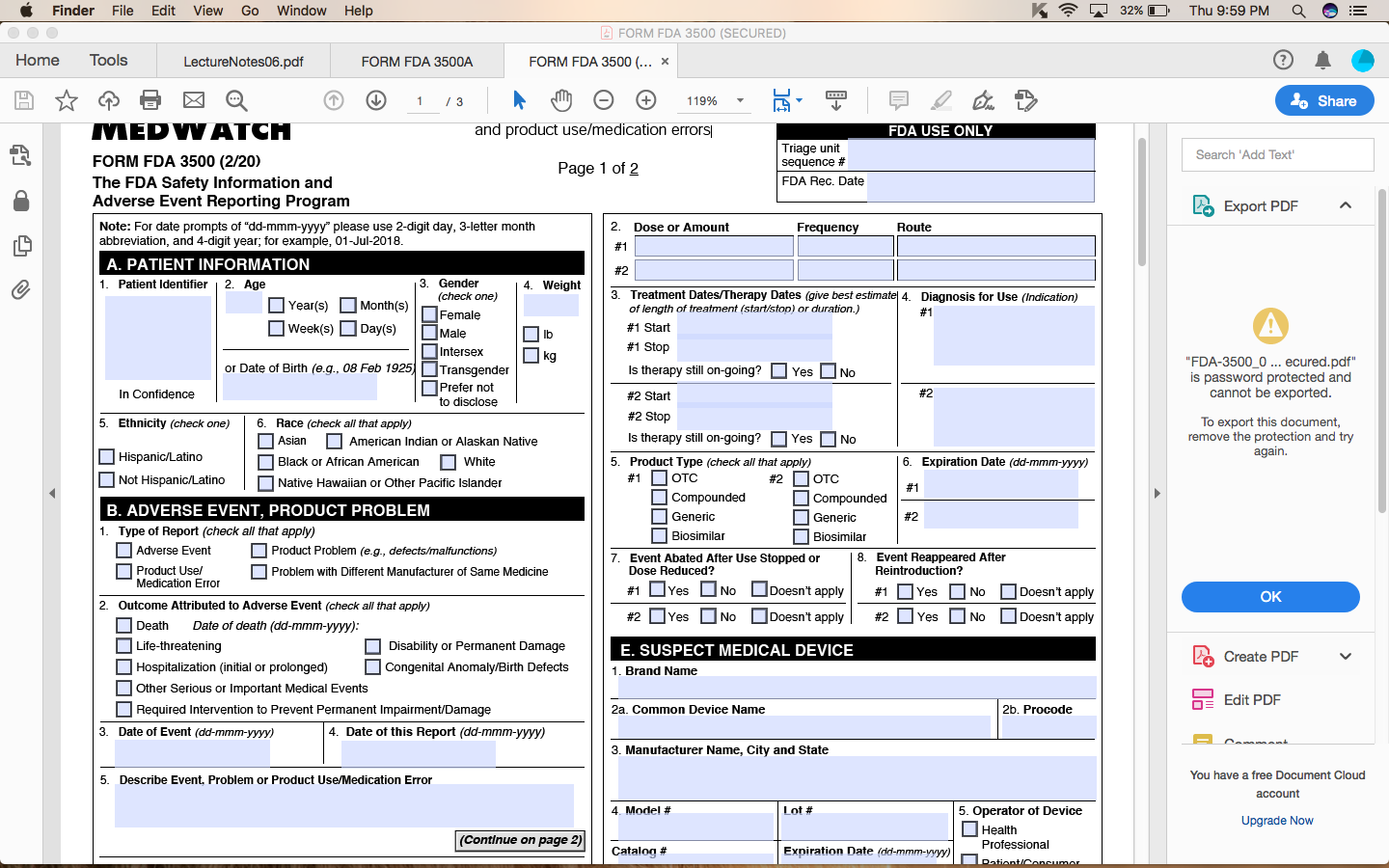
Form Fda 3500A ≡ Fill Out Printable PDF Forms Online
Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or.
PPT Structured Data Capture (SDC) Patient Safety Event & Adverse
Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every.
Instructions Using the information below
Fda medwatch offers several ways to help you stay informed about the medical products you prescribe, administer, or dispense every day: Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or.
Medwatch form 3500a Fill out & sign online DocHub
An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or. Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or.
Fda Medwatch Offers Several Ways To Help You Stay Informed About The Medical Products You Prescribe, Administer, Or Dispense Every Day:
Use medwatch to report a serious adverse event, product quality problem, product use error, therapeutic inequivalence/failure, or suspected. An important fda program called medwatch allows health care professionals and consumers to report serious problems that they believe may be. Reporting can be done through our online reporting portal or by downloading, completing and then submitting fda form 3500 (health professional) or.