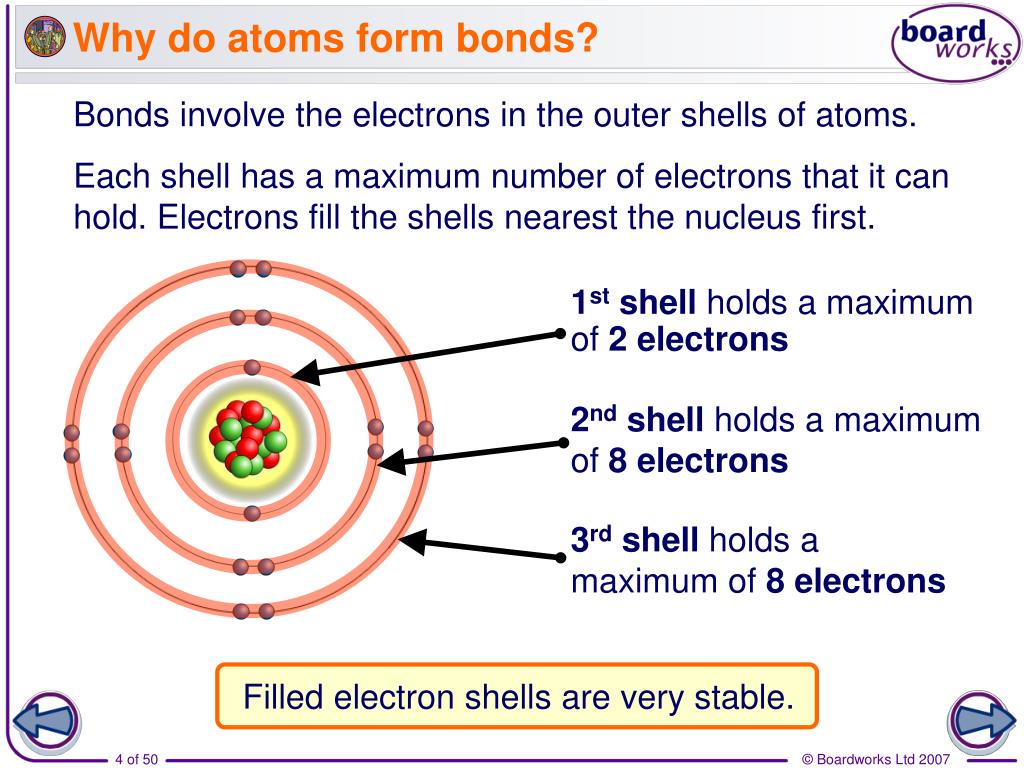
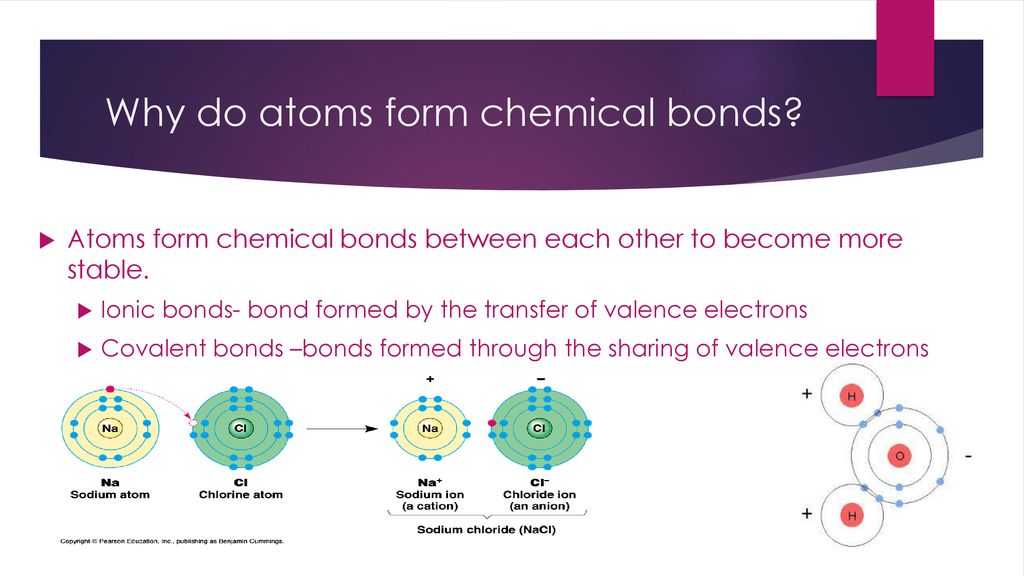
How Do Atoms Form Chemical Bonds - What do atoms do when they react with other atoms? Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. It contains four valence electrons and needs 4 more. This is because they're the most balanced and their outer. They form a molecule by the intermediate of chemical bonds. Noble gas atoms have a full valence shell of electrons, making them highly stable. Chemical bonding is very essential for atoms. This stability reduces the tendency for noble gases to. The noble gases are called that because they don't form bonds easily.
Noble gas atoms have a full valence shell of electrons, making them highly stable. The noble gases are called that because they don't form bonds easily. Chemical bonding is very essential for atoms. They form a molecule by the intermediate of chemical bonds. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. This is because they're the most balanced and their outer. It contains four valence electrons and needs 4 more. What do atoms do when they react with other atoms? This stability reduces the tendency for noble gases to.
What do atoms do when they react with other atoms? The noble gases are called that because they don't form bonds easily. Chemical bonding is very essential for atoms. It contains four valence electrons and needs 4 more. They form a molecule by the intermediate of chemical bonds. This stability reduces the tendency for noble gases to. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. Noble gas atoms have a full valence shell of electrons, making them highly stable. This is because they're the most balanced and their outer.
Compounds Thursday, 20 September ppt download
Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. The noble gases are called that because they don't form bonds easily. It contains four valence electrons and needs 4 more. They form a molecule by the intermediate of chemical bonds. What do atoms do when they react with other.
why do atoms form chemical bonds Mistery Rules
This is because they're the most balanced and their outer. They form a molecule by the intermediate of chemical bonds. This stability reduces the tendency for noble gases to. What do atoms do when they react with other atoms? Noble gas atoms have a full valence shell of electrons, making them highly stable.
PPT What are bonds? PowerPoint Presentation, free download ID5980343
They form a molecule by the intermediate of chemical bonds. This is because they're the most balanced and their outer. The noble gases are called that because they don't form bonds easily. This stability reduces the tendency for noble gases to. Noble gas atoms have a full valence shell of electrons, making them highly stable.
Why do atoms form chemical bonds Online Chemistry Lecture 9th Class
Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. Noble gas atoms have a full valence shell of electrons, making them highly stable. The noble gases are called that because they don't form bonds easily. It contains four valence electrons and needs 4 more. This is because they're the.
Chemistry of Life Chapter ppt download
This is because they're the most balanced and their outer. It contains four valence electrons and needs 4 more. The noble gases are called that because they don't form bonds easily. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. They form a molecule by the intermediate of chemical.
Chapter 8 Concepts of Chemical Bonding ppt download
Chemical bonding is very essential for atoms. The noble gases are called that because they don't form bonds easily. This stability reduces the tendency for noble gases to. They form a molecule by the intermediate of chemical bonds. Noble gas atoms have a full valence shell of electrons, making them highly stable.
Do Now Why do atoms form chemical bonds, and what happens during the
Chemical bonding is very essential for atoms. This stability reduces the tendency for noble gases to. It contains four valence electrons and needs 4 more. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. What do atoms do when they react with other atoms?
Types of Chemical Bonds
Noble gas atoms have a full valence shell of electrons, making them highly stable. It contains four valence electrons and needs 4 more. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. The noble gases are called that because they don't form bonds easily. They form a molecule by.
Aim What are covalent bonds and how are they formed? ppt download
They form a molecule by the intermediate of chemical bonds. Chemical bonding is very essential for atoms. This stability reduces the tendency for noble gases to. The noble gases are called that because they don't form bonds easily. What do atoms do when they react with other atoms?
Intro to Chemical Bonding ppt video online download
This is because they're the most balanced and their outer. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. It contains four valence electrons and needs 4 more. Noble gas atoms have a full valence shell of electrons, making them highly stable. Chemical bonding is very essential for atoms.
This Is Because They're The Most Balanced And Their Outer.
It contains four valence electrons and needs 4 more. They form a molecule by the intermediate of chemical bonds. Atoms of halogens, such as chlorine and fluorine, do form chemical bonds with other elements to achieve a stable electron. This stability reduces the tendency for noble gases to.
Chemical Bonding Is Very Essential For Atoms.
What do atoms do when they react with other atoms? Noble gas atoms have a full valence shell of electrons, making them highly stable. The noble gases are called that because they don't form bonds easily.